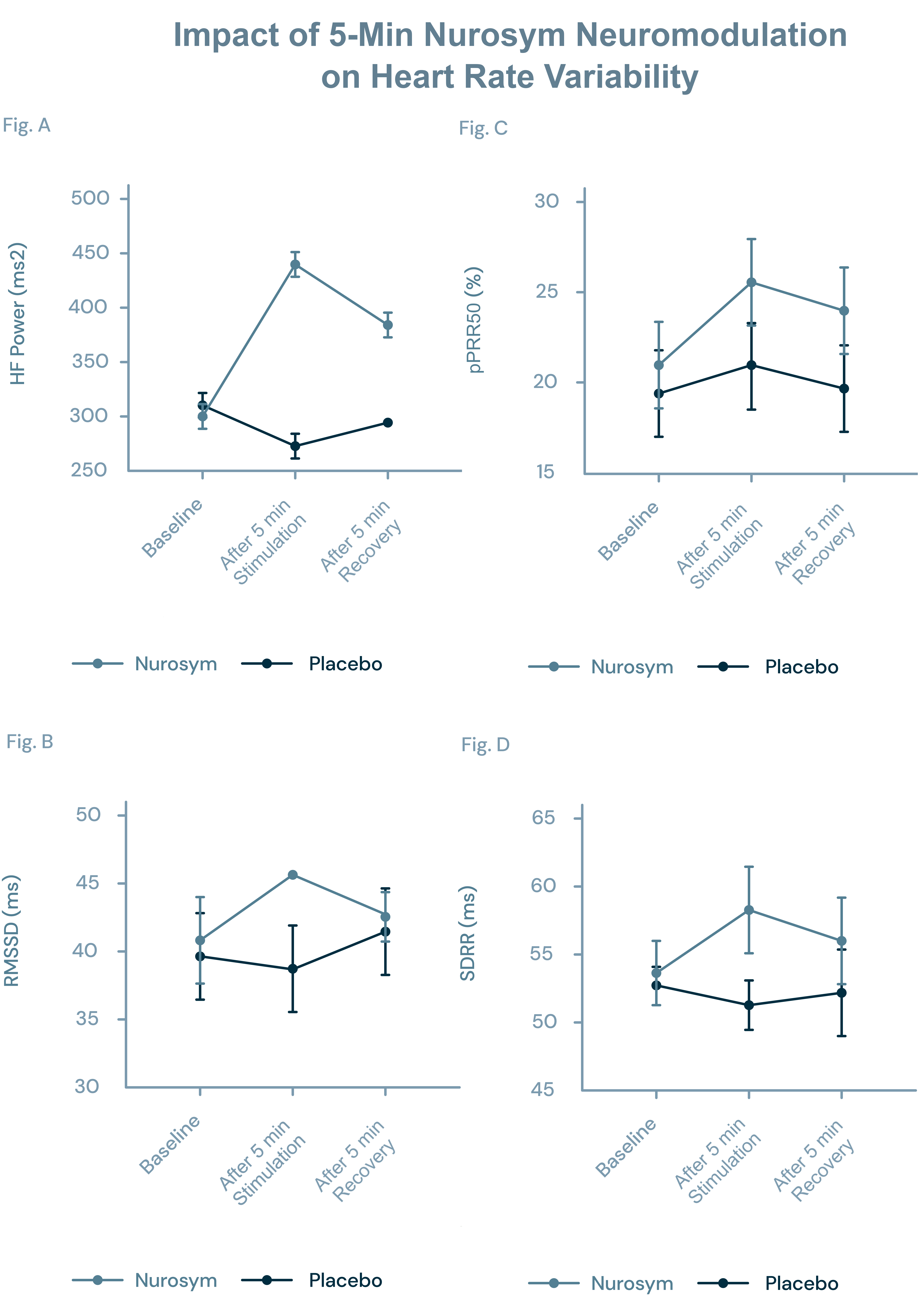

Parasym Clinical Trial, 2022
This study included 20 healthy participants. Participants abstained from caffeine, exercise, smoke, and alcohol in the 5 hours preceding the experiment and were blinded to active or sham treatment.
Autonomic function measured by HRV components was significantly affected in the active group (n=10) receiving Parasym neuromodulation compared to the placebo group (n=10) receiving placebo neuromodulation. After 5 minutes of neuromodulation the following HRV components were positively altered in the active group receiving Parasym neuromodulation compared to the placebo group.
(A) HF (High-Frequency Power)
(B) RMSSD (Root Mean Square of Successive Differences)
(C) pRR50 (Percentage of Successive RR Intervals > 50 ms Difference)
(D) SDRR (Standard Deviation of RR Intervals)
Changes HF, RMSSD, pRR50, and SDRR compared to the baseline persisted in the active group during the recovery phase, 5 minutes after the neuromodulation was interrupted. No adverse effects were observed by any of the participants.






